Find freezing point of different substance like freezing point of water hydrogen carbon nitrogen sodium aluminum iron zinc helium silver gold mercury lead iodine platinum and many more. Why does the Fahrenheit scale use 32 degrees as a freezing point.
 Temperature And Temperature Scales Chemistry For Non Majors
Temperature And Temperature Scales Chemistry For Non Majors
He called the temperature of an icesaltwater mixture zero degrees as this was the lowest temperature he could conveniently attain in his lab.

What is freezing point in fahrenheit. This is far lower than that of water. On the Fahrenheit scale however freezing is 32 degrees and boiling 212. The freezing point of water is 32F 0C.
3 rânduri Fahrenheit is a temperature scale that bases the boiling point of water at 212 and the. Get the answers you need now. Because oil is a petroleum product it will not freeze solid in cold temperatures though it will lose its liquid qualities as temperatures fall.
To get rid of the awkward fractions Fahrenheit did some multiplication eventually winding up with 32 as the freezing point and 96 as body temperature. Do 0 and 100 degrees Fahrenheit mean anything. For example diesel oil that is cooled below 0 degrees Fahrenheit will change in color and texture becoming whitish and solid to the point that it will not flow through the engine.
Do 0 and 100 degrees Fahrenheit mean anything. The temperature may be lower if supercooling occurs or if there are impurities present in the water which could cause freezing point depression to occur. The 18th-century German physicist Daniel Gabriel Fahrenheit originally took as the zero of his scale the temperature of an equal ice-salt.
Thats not always the case though. At room temperature mercury is a liquid. On the Fahrenheit scale the freezing point of water is 32 F and the boiling point is 212F at standard atmospheric pressure.
Fahrenheit temperature scale scale based on 32 for the freezing point of water and 212 for the boiling point of water the interval between the two being divided into 180 equal parts. According to Jefferson Labs mercurys melting and freezing point is -388 degrees Celsius. What is freezing point of water in degrees FahrenheitI want full solution.
Its boiling point is 3567 degrees Celsius 6299 Kelvin or 6741 degrees Fahrenheit far higher than water. Everybody knows 0 degrees on the Celsius scale is the freezing point of water and 100 degrees is the boiling point. You might be interested.
I want full solution. For example by adding salt sodium chloride to water which will freeze at 32F 0C t. First of all you must know that the freezing point of liquids is determined both by the temperature of the liquid AND the contents in solution of the liquid.
Scientists have found liquid water as cold as. SVMishra SVMishra 04022021 Math Primary School answered What is freezing point of water in degrees Fahrenheit. On the Fahrenheit scale however freezing is 32 degrees and boiling 212.
Under certain conditions water may remain a liquid as cold as -40 to -42F. Freezing point definition the temperature at which a liquid freezes. How on earth were these numbers arrived at.
Ordinarily the freezing point of water and melting point is 0 C or 32 F. Maria Ciaramitaro Answer Daniel Fahrenheit did not use the freezing point of water as a basis for developing his scale. Weve all been taught that water freezes at 32 degrees Fahrenheit 0 degrees Celsius 27315 Kelvin.
How on earth were these numbers arrived at. This is the same as 2343 Kelvin and -379 degrees Fahrenheit. Everybody knows 0 degrees on the Celsius scale is the freezing point of water and 100 degrees is the boiling point.
Freezing point of Boron B is 2076 C Convert 2076 C to different units. 2 See answers.
Accordingly what is the freezing point for Kelvin. Kelvin is a unit of measurement of temperature.
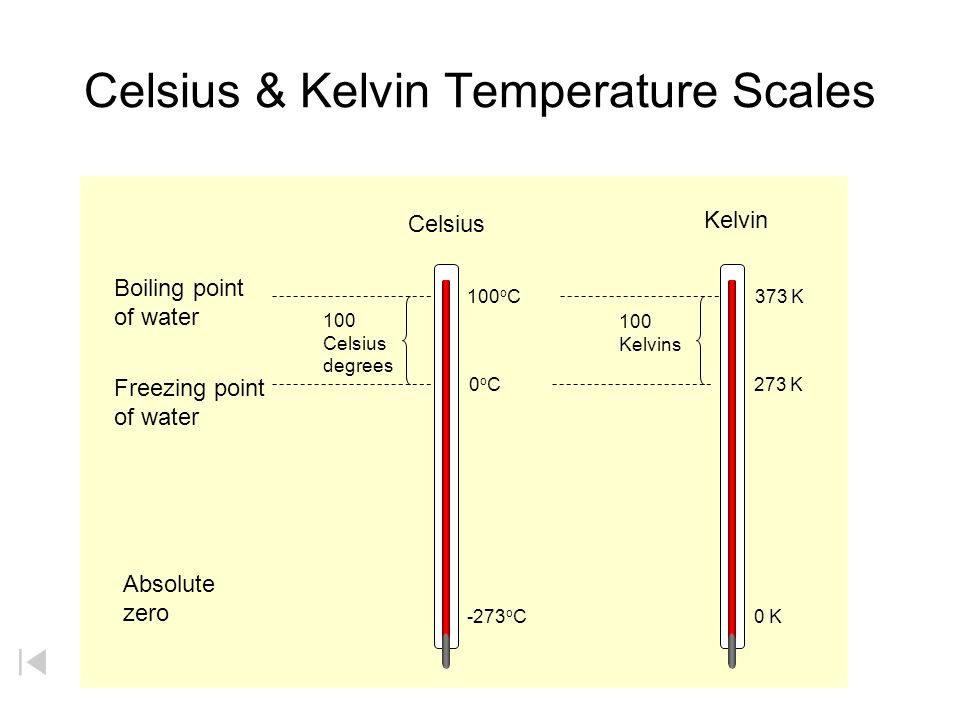 Temperature V Heat Celsius Kelvin Temperature Scales Boiling Point Of Water Freezing Point Of Water Absolute Zero Celsius 100 Celsius Degrees Ppt Download
Temperature V Heat Celsius Kelvin Temperature Scales Boiling Point Of Water Freezing Point Of Water Absolute Zero Celsius 100 Celsius Degrees Ppt Download
Hence T02731527315Kfreezing point of water in Kelvin scale Answered By.

Freezing point of kelvin. Absolute zero is 27315 C or 45967 F. Because at 273 K the temperature in centigrades is 0 which is the normal freezing point of water. Liquid nitrogen is not colder than liquid helium.
Water boils at a temperature of 100 C which is 37315 kelvins or 212 F. K f molal freezing point depression constant or cryoscopic constant in C kgmol. Kelvin Celsius 27315 therefore assuming you are referring to the freezing point of water 0C freezing point 27315K.
Molality m of NaCl moles of NaClkg water. Is Liquid Nitrogen colder. The size of a one degree change in temperature is exactly the same in the Celsius and Kelvin scales so the freezing point of water is at a temperature of 27315 kelvins that is 27315 degrees above absolute zero.
It also happens to be the only material that doesnt exist as a solid it. This very low freezing point is a characteristic of all noble gases. Mechanics Forces of Freezing Water Effects of ice and.
The Freezing point of water in kelvin. M molality of the solute in mol solutekg solvent. The freezing point of water in degree Celsius is 0C.
The others are helium argon krypton xenon and radon. This is the same as 2343 Kelvin and -379 degrees Fahrenheit. Atomic mass Na 2299.
The freezingmelting point of water is about 27315 K at a pressure of 1 atmosphere. The Kelvin degree is the same size as the Celsius degree. The simple fact is that that liquid helium is the coldest known material as it has a boiling point of -452F.
Absolute zero is 27315 C or 45967 F. The Kelvin degree is the same size as the Celsius degree. Kelvin to Celsius conversion.
50 Freezing Point Of Kelvin Images. Its boiling point is 3567 degrees Celsius 6299 Kelvin or 6741 degrees Fahrenheit far higher than water. So basically both temperatures are the same and.
Absolute zero is the lowest limit of the thermodynamic temperature scale a state at which the enthalpy and entropy of a cooled ideal gas reach their minimum value taken as zero kelvinsThe fundamental particles of nature have minimum vibrational motion retaining only quantum mechanical zero-point energy-induced particle motionThe theoretical temperature is determined by extrapolating the. Step 1 Calculate the molality of the NaCl. The Kelvin degree is the same size as the Celsius degree.
Hence the two reference temperatures for Celsius the freezing point of water 0C and the boiling point of water 100C correspond to 27315K and 37315K respectively. Hence the two reference temperatures for Celsius the freezing point of water 0C and the boiling point of water 100C correspond to 27315K and 37315K respectively. Temperature scale that registers the freezing.
The size of a one degree change in temperature is exactly the same in the Celsius and Kelvin scales so the freezing point of water is at a temperature of 27315 kelvins that is. According to Jefferson Labs mercurys melting and freezing point is -388 degrees Celsius. Melting points in blue and boiling points in pink of the first eight carboxylic acids C For most substances melting and freezing points are approximately equal.
The symbol of Kelvin is K. From the periodic table find the atomic masses of the elements. The freezing point of neon is minus 4155 degrees Fahrenheit minus 2486 degrees Celsius which is only slightly lower than its boiling point of minus 41066 F minus 24592 C.
This is also its melting point. The freezing point of water on Celsius scale is 0oCWe have TKtoC27315. For example the melting point and freezing point of mercury is 23432 kelvins 3883 C.
Hence the two reference temperatures for Celsius the freezing point of water 0C and the boiling point of water 100C correspond to 27315K and 37315K respectively. This zero point motion is significant enough that it prevents the atoms from sticking together as a solid. It does however have its lowest point as absolute zero the lowest possible temperature when all atoms will have no kinetic energy and so temperature can not get any lower.
Neon is one of six noble gases. This is far lower than that of water. This is why Helium doesnt freeze at absolute zero.
Kf is the freezing point depression constant. In other words it was upside-down.
/the-freezing-point-of-water-609418_FINAL-01f50f5f4f7d4a39854bebcc59df1aa4.gif) What Is The Freezing Point Of Water
What Is The Freezing Point Of Water
Sign up for the The scale places the freezing and boiling points of water exactly 180 degrees apart because in this scale the freezing point of water is 32 degrees 32 F and the boiling point is 212 degrees.

Freezing point and boiling point of water. The following equation is used to calculate the freezing point of a liquid. Because the freezing point of pure water is 0C the sucrose solution freezes at 068C. This Demonstration shows the boiling temperature of water on six planets all of which have different atmospheric pressures.
One at room temperature 72F22C one at the same temperature as my hot water tap 115F46C and one boiling 212F100C. Since liquid salt decreases the freezing point of water the temperature of the bag of salt drops below zero degrees -1 degree Celsius. This boiling-point difference of 161 millikelvins between the Celsius scales original definition and the previous one based on absolute zero and the triple point has little practical meaning in common daily applications because waters boiling point is very sensitive to variations in barometric pressure.
At 10000 feet above sea level the pressure of the atmosphere is only 526 mmHg. I started with three identical glass containers each holding 100ml about 35 fl. M is the molality.
A solution boils at a slightly higher temperature than the pure solvent. How to use freezing point in a sentence. The change in the boiling point is calculated from.
When the opposite happens and a liquid turns into a solid it is called freezing. When a liquid becomes a gas it is called boiling or vaporization. About Press Copyright Contact us Creators Advertise Developers Terms Privacy Policy Safety How YouTube works Test new features Press Copyright Contact us Creators.
Thus as solutes or solids are applied to liquids the freezing point drops and the boiling point rises. Click to see full answer. A similar property of solutions is boiling point elevation.
The normal boiling point of water is 100 o C because this is the temperature at which the vapor pressure of water is 760 mmHg or 1 atm. Note that ceCaCl_2 is substantially more effective at lowering the freezing point of water because its solutions contain three ions per formula unit. A freezing point is the temperature at which a liquid transitions to a solid.
The red line shows the atmospheric pressure of the selected planet. In Celsius the boiling and freezing points of water are 100 and 0 respectively. Therefore the boiling point elevation would be 2oC.
In Fahrenheit those would be 212 and 32. T K f m. Because the freezing point of pure water is 0C the actual freezing points of the solutions are 22C and 30C respectively.
I knew that the water would not turn from liquid to ice all at once and I puzzled over when to consider the water. Asked May 7 2018 in Physics by paayal 147k points The efficiency of an ideal heat engine working between the freezing point and boiling point of water is -. The freezing point depression is the amount that the freezing temperature decreases.
For example an altitude change of only 28 cm 11 in causes the boiling point. About Press Copyright Contact us Creators Advertise Developers Terms Privacy Policy Safety How YouTube works Test new features Press Copyright Contact us Creators. He came up with this temperature scale based on the three fixed temperature points of the human body freezing water and a mixture of ice salt ammonium chloride and water.
The Fahrenheit scale defines the freezing point of water as 32F and the boiling point as 212F. The blue line shows the vapor pressure of water as a function of temperature. Under normal conditions when the pressure of the atmosphere is approximately 760 mmHg water boils at 100 o C.
The bag of the salt became colder so the mixture froze faster. The boiling point is the temperature at which the vapor pressure is equal to the atmospheric pressure. The melting point for water is 0 degrees C 32 degrees F.
For example the boiling point of pure water at 10atm is 100oC while the boiling point of a 2 saltwater solution is about 102oC. Freezing point definition is - the temperature at which a liquid solidifies. Oz of filtered water.
The freezing point of water is the same as its a. I put all these into my freezer which had an air temperature of 0F 18C. In fact ceCaCl_2 is the salt usually sold for home use and it is also often.
Where T is the freezing point. The Celsius scale sets the freezing point and boiling point of water at 0C and 100C respectively.